Fleming Polymer Ltd
Fleming Polymer Testing & Consultancy is an independent test-house offering the outsourcing of specialised analytical polymer testing facilities to the polymer industry. The business is dedicated to solving polymer problems through sophisticated testing, high level consultancy and process simulation. Our customers, most of whom are household names, are of course the cornerstone of the business, and to this end we appreciate the need to provide rapid solutions.
We deliver value added benefits, which serve both to protect and increase profitability and operate to ISO 9001:2008. The company has been operating for over 15 years, has won a DTI Smart award and been granted a patent for rheometry design.
The company has worked with industry leaders such as; GlaxoSmithKline, BD, Exxon, Uponor, Malvern Instruments, Tyco, TIAutomotive, NPL, Moldflow and Victrex to name but a few. Test Specialisms include;
- Capillary Rheometry
- FTIR
- Extrusion Flow Simulation
- MFR/MVR
- DSC
- TGA
- Instrumented Impact
- Elongational Viscosity
- Compuplast VEL distributor
Capillary Rheometry
Most of today's polymer processing operations exploit deformation and flow in order to create the vast range of polymeric products available. A rheometer replicates the deformation and flow regimes typically experienced in extrusion and injection moulding; the ability to replicate the flow remotely is instrumental in process optimisation and material characterisation.
Rheometry is historically associated with viscosity measurement, and whilst this is an important rheological parameter it constitutes only one of a number that are required to yield a comprehensive characterisation. Some of the other essential parameters are: shear viscosity,shear stress, shear rate, extensional viscosity, extensional stress, extensional strain, melt fracture, wall slip and elasticity.

FTIR
Fleming PTC has recently expanded its service portfolio to include the technique of FTIR. To this end a Spectrum Two with ATR has been purchased from industry leader Perkin Elmer and has rapidly confirmed itself as a complementary and staple polymer characterisation test.

Thermal Analysis
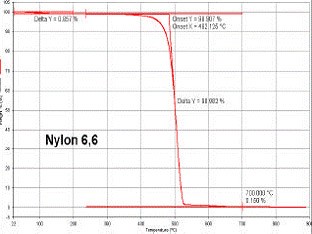
Thermal analysis techniques provided are both DSC and TGA. DSC has provision for the determination of melting and crystallisation temperatures, heat of fusion, Tg, OIT and specific heat capacity. Nitrogen and argon purge gases are available for ambient and sub-ambient conditions. TGA has the provision for gas switching from either inert to oxygen or air.

Flow simulation
Why Flow Simulation? The simulation of polymer flow processes is extremely important because it allows us the opportunity to eliminate the need for costly trials using real materials and real people on real plant. A range of process scenarios can be created in a virtual environment and conclusions drawn as to their suitability in minutes rather than days or weeks. Compuplast provide software for almost every extrusion process; spiral mandrel, profile, pipe, film and sheet. Furthermore, the software also has a single screw extruder simulation module which allows the investigation of the extrusion process from complex screw design to barrel temperature and screw speed optimization.
Extrusion
A Betol 25mm diameter single screw extruder is available for small scale extrusions and compounding. It is often required to blend a polymer sample for either using in another extrusion technique and/or investigating the rheological behaviour of the blend using offline instruments such as the capillary rheometer. However, the rheometer is not capable of mixing two or more materials and assumes a totally homogeneous sample is being tested. When this is the case, a blend can be compounded on the extruder, pelletized and the product used in the off-line techniques. The Betol 25mm extruder with its water bath and pelletizer is perfect for these operations as well as driving the film tower.

Pilot blown film line
A lab- scale blown film line is available for the pilot production of developmental mono-layer film formulation and trials. In many cases the extruder can be used beforehand to create a blend which can then be extruded again using the film line. In this way a series of formulations can be examined for process optimisation and service properties on a small scale rather than the costlier alternative of using a production-scale line.

Melt Mass-Flow Rate
The Melt Mass-Flow Rate is a one number indication of the viscosity of a polymer in the melt phase. It is defined as the mass of polymer in grams flowing per 10 minutes through a capillary of specific diameter and length by a pressure applied via a range of standard weights at specified temperature.
MFR is a simple test and is useful in that a feel for polymer viscosity can be attained very quickly. Furthermore, the MFI value is indicative of polymer weight average molecular weight and, if using the ratio of two MFR values from dissimilar weights (flow rate ratio-FRR), its molecular weight distribution.
The test also lends itself to the determination of melt density, which is an essential parameter in an overall characterisation.
MFR constitutes one of the most frequently used tests in the polymer converting industry to control incoming material quality, since it is quick and requires very little operator training to return representative results. Some companies prefer the use of the melt volume rate (MVR) instead of the MFR value.
The MVR is reported in cm3/10 mins; the only difference being that MFR, a mass flow rate, requires the density to be known beforehand.
The method is detailed in the following two well known standards: ASTM D1238 and ISO 1133.
FlemingPTC uses the CEAST model 6942 (see picture) with automatic weight lifting and maximum temperature of 400ºC. Masses available include; 2.16Kg, 5Kg, 10Kg and 21.6Kg. Die diameter, 2.095mm; die length 8mm. The model is designed to adhere to the following standards:
- ISO1133
- ASTM D1238 methods A and B, D3364
- DIN 53735
- BSI 2782 method 720A
- UNI 5640
- JIS K 7210
- AFNOR NF T 51-016
The melt flow rate is an indirect measure of molecular weight, high melt flow rate corresponding to low molecular weight. At the same time, the melt flow rate is a measure of the ability of the material's melt to flow under pressure.
The melt flow rate is inversely proportional to the viscosity of the melt at the conditions of the test, though it should be born in mind that the viscosity for any such material depends on the applied force.
Ratios between two melt flow rate values (FRR) for one material at different gravimetric weights is often used as an indication the broadness of the molecular weight distribution.
Melt flow rate is very commonly used for polyolefins, polyethylene being measured at 190°C and polypropylene at 230°C.
The plastics converter should ideally choose a material with a melt index high enough to easily form the polymer into the article intended, but on the other hand, low enough that the mechanical strength of the final article will be sufficient for its use.

Instrumented impact testing
Instrumented impact testing today constitutes and important end product and fit for purpose test. Following manufacture many products will experience some form of service impact specific to their environment, it is the aim of the impact tester to replicate these service impacts in order to assess their fitness for purpose. Instrumented impact, as opposed to pendulum impact, generates the entire force vs time fingerprint and provides a more thorough insight into the particular failure mode.

Polymer elasticity inferred by die swell
Fleming PTC has added a laser die swell system to its Malvern Instruments Rosand RH7 twin bore capillary rheometer. The system allows the accurate and continuous measurement of polymer die swell, which is an important rheological measurement, instrumental in the understanding of elasticity. Elasticity is important in polymer extrusion, since most polymers in the melt phase are viscoelastic. Such materials will swell to some degree on exiting a production die; a problem if you want your extrusion to share the dimensions of your die! Melted polymers, in most cases, experience stretching through extrusion dies, and due to the restoring forces generated, will try to return to their original state on exit (swelling). The magnitude of the restoring forces defines the elasticity, which can be hard to measure in an absolute fashion (first normal stress difference, N1), however, die swell, is a convenient way to infer the elasticity; the more the swell the more elastic the polymer. In this way, screening of various grades can predict which are more elastic and which will swell more. Once elasticity is inferred, a designer can work on either refining the die design and/or process conditions to achieve the desired degree of swell, or the grade could be switched altogether in favour of a more suitable alternative. In either case, knowledge of this important rheological parameter allows choices to be made which may otherwise appear impossible. The measurement is described in ISO11443:2005, 8.7 Determination of extrudate swelling.

Shear Viscosity
Bagley and Rabinowitsch corrected, essential for result integrity and pre-requisite for most die design and flow simulation packages.

Rheology Training and Interpretation
Rheometry training is essential if the best use is to be made of what are very complex instruments. Many field leaders are requesting assistance with tuition on both rheometry fundamentals and the more sophisticated techniques such as wall slip, elongational viscosity and elasticity.
A highly sophisticated technique such as rheometry can often see instruments unused for enough time that operators forget how to use the machine or, personnel leave and new operators have to get up to speed. In both scenarios courses can be tailored to train or retrain key personnel to the level required.
Melt Volume-Flow Rate
Summarising, MFR/MVR is:
- An easy to use 1 number test
- Indicative of weight average molecular weight
- Indicative of polymer melt viscosity
- Indicative of molecular weight distribution
- An easy to implement quality test
- Easy to implement incoming material acceptance test
- Essentially a simple capillary rheometer

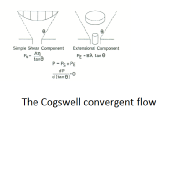
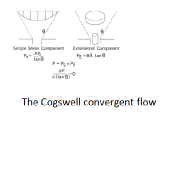
Elongational Viscosity
Determined via the Cogswell convergent flow model. Elongational viscosity, or the resistance to elongation is known to be sensitive to material structure (MWD and chain branching).
Dissimilar MWD can often be resolved in radioactive and peroxide cross-linked LLDPEs that other techniques (shear viscosity and MFI) are blind to. Branching differences, such as those seen in HDPE and LDPE, can also be differentiated as can changes due to catalyst type using this measurement.
Wall Slip
Slip flow in the processing of polymers may be either beneficial, and actively encouraged by the addition of process additives, or detrimental, for example relating to the occurrence of flow instabilities. However, the occurrence of slip results in complications in both the measurement and modelling of the flow behaviour of materials. Reliable characterisation of the rheological behaviour of materials is important; whether it is for developing new materials with specific flow properties, design of injection moulding and extrusion processes, or for quality control of materials. This measurement is available via capillary rheometry and uses the Mooney method for the calculation of slip velocities from the experimental data.
Differential Scanning Calorimetry
Differential Scanning Calorimetry (DSC) is widely used to characterise the thermophysical properties of polymers. DSC can measure important thermoplastic properties including:
- Melting temperature
- Heat of melting
- Percent crystallinity
- Tg or softening
- Crystallisation
- Presence of recyclates/regrinds
- Plasticisers
- Polymer blends (presence, composition and compatibility
Differential scanning calorimetry or DSC is a thermoanalytical technique in which the difference in the amount of heat required to increase the temperature of a sample and reference are measured as a function of temperature. Both the sample and reference are maintained at very nearly the same temperature throughout the experiment.
Generally, the temperature program for a DSC analysis is designed such that the sample holder temperature increases linearly as a function of time. The reference sample should have a well-defined heat capacity over the range of temperatures to be scanned.
The basic principle underlying this technique is that, when the sample undergoes a physical transformation such as phase transitions, more (or less) heat will need to flow to it than the reference to maintain both at the same temperature.
Whether more or less heat must flow to the sample depends on whether the process is exothermic or endothermic. For example, as a solid sample melts to a liquid it will require more heat flowing to the sample to increase its temperature at the same rate as the reference.
This is due to the absorption of heat by the sample as it undergoes the endothermic phase transition from solid to liquid. Likewise, as the sample undergoes exothermic processes (such as crystallisation) less heat is required to raise the sample temperature. By observing the difference in heat flow between the sample and reference, differential scanning calorimeters are able to measure the amount of heat absorbed or released during such transitions.
DSC may also be used to observe more subtle phase changes, such as glass transitions. DSC is widely used in industrial settings as a quality control instrument due to its applicability in evaluating sample purity and for studying polymer curing. The result of a DSC experiment is a heating or cooling curve.
This curve can be used to calculate enthalpies of transitions. This is done by integrating the peak corresponding to a given transition. It can be shown that the enthalpy of transition can be expressed using the following equation:
ΔH = KA
where ΔH is the enthalpy of transition, K is the calorimetric constant, and A is the area under the curve. The calometric constant will vary from instrument to instrument, and can be determined by analysing a well-characterised sample with known enthalpies of transition.
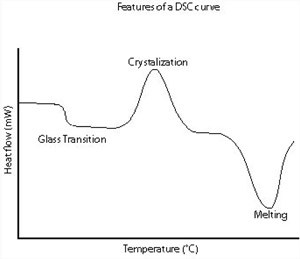
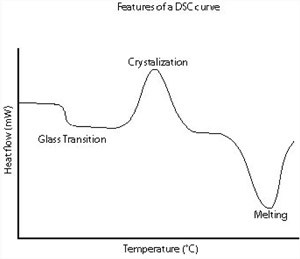
A schematic DSC curve demonstrating the appearance of several common features.
Differential scanning calorimetry can be used to measure a number of characteristic properties of a sample. Using this technique it is possible to observe fusion and crystallisation events as well as glass transition temperatures (Tg). DSC can also be used to study oxidation, as well as other chemical reactions.
Glass transitions may occur as the temperature of an amorphous solid is increased. These transitions appear as a step in the baseline of the recorded DSC signal. This is due to the sample undergoing a change in heat capacity; no formal phase change occurs. As the temperature increases, an amorphous solid will become less viscous.
At some point the molecules may obtain enough freedom of motion to spontaneously arrange themselves into a crystalline form. This is known as the crystallisation temperature (Tc). This transition from amorphous solid to crystalline solid is an exothermic process, and results in a peak in the DSC signal.
As the temperature increases the sample eventually reaches its melting temperature (Tm). The melting process results in an endothermic peak in the DSC curve. The ability to determine transition temperatures and enthalpies makes DSC an invaluable tool in producing phase diagrams for various chemical systems.
Using differential scanning calorimetry to study the oxidative stability of samples generally requires an airtight sample chamber. Usually, such tests are done isothermally (at constant temperature) by changing the atmosphere of the sample. First, the sample is brought to the desired test temperature under an inert atmosphere, usually nitrogen.
Then, oxygen is added to the system. Any oxidation that occurs is observed as a deviation in the baseline. Such analyses can be used to determine the stability and optimum storage conditions for a compound.
DSC is widely used in the pharmaceutical and polymer industries. For the polymer chemist, DSC is a handy tool for studying curing processes, which allows the fine tuning of polymer properties. The cross-linking of polymer molecules that occurs in the curing process is exothermic, resulting in a positive peak in the DSC curve that usually appears soon after the glass transition.
In the pharmaceutical industry it is necessary to have well-characterised drug compounds in order to define processing parameters. For instance, if it is necessary to deliver a drug in the amorphous form, it is desirable to process the drug at temperatures below those at which crystallisation can occur.
In food science research, DSC is used in conjunction with other thermal analytical techniques to determine water dynamics. Changes in water distribution may be correlated with changes in texture. Similar to material science studies, the effects of curing on confectionery products can also be analysed.
DSC curves may also be used to evaluate drug and polymer purities. This is possible since the temperature range over which a mixture of compounds melts is dependent on their relative amounts.
This effect is due to a phenomenon known as freezing point depression, which occurs when a foreign solute is added to a solution. Consequently, less pure compounds will exhibit a broadened melting peak that begins at lower temperature than a pure compound.
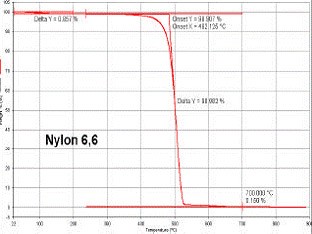
Thermo-Gravimetric Analysis
Thermogravimetric analysis (TGA) is one of the members of the family of thermal analysis techniques used to characterise a wide variety of materials.
TGA provides complimentary and supplementary characterisation information to the most commonly used thermal technique, DSC.
TGA measures the amount and rate (velocity) of change in the mass of a sample as a function of temperature or time in a controlled atmosphere. The measurements are used primarily to determine the thermal and/or oxidative stabilities of materials as well as their compositional properties.
The technique can analyse materials that exhibit either mass loss or gain due to decomposition, oxidation or loss of volatiles (such as moisture). It is especially useful for the study of polymeric materials, including thermoplastics, thermosets, elastomers, composites, films, fibres, coatings and paints.
TGA measurements provide valuable information that can be used to select materials for certain end-use applications, predict product performance and improve product quality. The technique is particularly useful for the following types of measurements:
The Perkin Elmer TGA7
- Compositional analysis of multi-component materials or blends
- Thermal stabilities
- Oxidative stabilities
- Estimation of product lifetimes
- Decomposition kinetics
- Effects of reactive atmospheres on materials
- Filler content of materials
- Moisture and volatiles content
Melt Fracture
a very real problem in film blowing, sheet and pipe extrusion manifesting itself as a surface distortion or sharkskin. Melt fracture is easily detected as pressure fluctuations in capillary rheometry, thus enabling the onset of fracture to be predicted off-line using a small quantity of material.

Flow Visualisation
rheological characterisation is increasingly being used in flow simulation software such as Compuplast for die\screw design, multi-layer extrusion and film blowing optimisation.
Pressure Calibration
- Pressure range: 0.1MPa to 70MPa
- Dual area piston with automatic changeover
Allows highly accurate calibration of pressure transducers. Essential for rheometry since stress and all subsequent calculations are based on pressure. It can be difficult to tell when a pressure transducer is damaged and reading incorrectly without comparing against an known reference. Regular checking and calibration is recommended in order to ensure result integrity.